EYE / FACIAL PROTECTION
What are common types of eye protection?
Goggles
Appropriately fitted, indirectly-vented goggles* with a manufacturer’s anti-fog coating provide the most reliable practical eye protection from splashes, sprays, and respiratory droplets. Newer styles of goggles may provide better indirect airflow properties to reduce fogging, as well as better peripheral vision and more size options for fitting goggles to different workers. Many styles of goggles fit adequately over prescription glasses with minimal gaps. However, to be efficacious, goggles must fit snugly, particularly from the corners of the eye across the brow. While highly effective as eye protection, goggles do not provide splash or spray protection to other parts of the face.
* Directly-vented goggles may allow penetration by splashes or sprays; therefore, indirectly-vented or non-vented goggles are preferred for infection control.
Face Shields
Face shields are commonly used as an infection control alternative to goggles.** As opposed to goggles, a face shield can also provide protection to other facial areas. To provide better face and eye protection from splashes and sprays, a face shield should have crown and chin protection and wrap around the face to the point of the ear, which reduces the likelihood that a splash could go around the edge of the shield and reach the eyes. Disposable face shields for medical personnel made of light weight films that are attached to a surgical mask or fit loosely around the face should not be relied upon as optimal protection.
** In a chemical exposure or industrial setting, faceshields should be used in addition to goggles, not as a substitute for goggles (ANSI Z87.1–2003 Practice for occupational and educational eye and face protection).
Safety Glasses
Safety glasses provide impact protection but do not provide the same level of splash or droplet protection as goggles and generally should not be used for infection control purposes.
Full-face Respirators
Full facepiece elastomeric respirators and powered air-purifying respirators (PAPRs) are designed and used for respiratory protection, but because of their design incidentally provide highly effective eye protection as well. Selection of this type of PPE should be based on an assessment of the respiratory hazard in an infection control situation, but will also provide, as an additional benefit, optimal eye protection.
Face shield design and structure
The majority of eye and face protection currently used in the U.S. is designed, tested, and manufactured in accordance with the American National Standards Institute (ANSI)/International Safety Equipment Association (ISEA) Z.87.1–2010 standard. The major structural components of a face shield include the following:
- Visor. Visors, also referred to as lenses or windows, are manufactured from any of several types of materials that include polycarbonate, propionate, acetate, polyvinyl chloride, and polyethylene terephthalate glycol (PETG) and come in disposable, reusable, and replaceable models . Acetate provides the best clarity and PETG tends to be the most economical, but polycarbonate is one of the most widely used. Polycarbonate and propionate offer better, although still somewhat imperfect, optical quality that aids in reducing eye strain associated with face shield wear. Visors can be treated with advanced coatings to impart anti-glare, anti-static, and anti-fogging properties, ultraviolet light (UV) protection, and scratch resistance features to extend the life of the visor. Some models come with built-in goggles that are incorporated into the visor. Visors are available in different lengths that include half facepiece length extending to the mid-face, full facepiece length that extends to the bottom of the chin, and a face/neck length that also covers the anterior neck area . Most visors curve around the face and come in different widths; wider visors offer more peripheral protection. Some one-piece face shields have visors that conform to the wearer’s face upon donning. Recommendations from the Centers for Disease Control and Prevention (CDC) are for visors that are of sufficient width to reach at least the point of the ear, as this will lessen the chances of the likelihood that a splash could go around the edge of the face shield and reach the eyes. In addition, visors should have crown and chin protection for improved infection control purposes. Some models of disposable medical/surgical face masks are available with an integral, thin plastic visor fitted to the top of the mask with an anti-fogging device between them to reduce the effects of exhaled moisture.
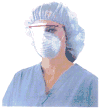
Figure 1 — Half face piece face shield with eyewear-like temple bars (Figure courtesy of the CDC).

Figure 2 — Frontal (a) and lateral (b) views of a reusable face/neck length face shield with brow cap, top band, and ratchet adjustment.
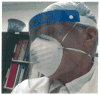
Figure 3 — Disposable one-piece face/neck length face shield visor assembly with foam forehead cushion and elastic strap.

Figure 4 — Surgical face mask with integral visor (Figure courtesy of Walter Reed National Military Medical Center).
2. Frame. Face shield frames used in healthcare are generally made of lightweight plastic. There are a variety of frame styles, including adjustable and nonadjustable frames that fully or partially encircle the circumference of the skull or those with eyeglass-type temple bars that are worn like standard eyewear . There are also metal clip-on frames available that are designed to attach face shield visors to prescription eyewear, and some frames allow for the visor to be flipped up when not in use. A number of manufacturers offer detachable frames for easy change-out of the face shield visor. Some models also incorporate a brow cap into the frame that affords additional splash protection in the forehead region, as well as allowing for more visor distance from the face that better accommodates the wearing of additional PPE (e.g., goggles, loupes, prescription eyewear, respirators) . Disposable visor-only face shields are also available that have a forehead foam cushion that provides a comfortable seal to the forehead .
3. Suspension Systems. There are a variety of face shield suspension systems on the market that offer either fully or partially circumferential attachment features. Fully circumferential suspension systems include plastic headbands that are adjustable for comfort by a ratchet mechanism, pin-lock systems, or Velcro®; nonadjustable systems employ elastic straps. Some models utilize eyeglass-type temple bars for suspension with or without eyewear-like nose pads and bridge assemblies to assist in maintaining face shield position and stability on the face. A top band that is adjusted for depth is found on some models.
Face shields provide a barrier to acutely-expelled aerosols of body fluids and are commonly used as an alternative to goggles as they confer protection to a larger area of the face. Utilizing a cough aerosol simulator loaded with influenza virus (aerosol volume mean diameter of 8.5 μm) and a breathing simulator, it is reported 96% and 92% reductions in the risk of inhalational exposure immediately after a cough for a face shield at distances of 18 in (46 cm) and 72 in (183 cm), respectively. Decreasing the aerosol size to 3.4 μm resulted in the face shield blocking 68% of the inhalational exposure at 18 in (46 cm) immediately after the cough and 23% over 1–30 min post-cough (during which time the larger aerosol particles had settled out and droplet nuclei had formed and remained airborne so that flow occurred more easily around the edges of the face shield). Other tests sprayed a fluorescent dye (particle diameter ~5μm) at a distance of 20 in (50 cm) away from a mannequin head outfitted with various types of PPE. They found that a face shield with head/neck length, three separate contact points at the forehead, and side curve reaching to the point of the ear, or the combination of this face shield and an N95 filtering facepiece respirator (N95 FFR) or any other FFR like KN95 , protected the eyes, nares and mouth from contamination. Conversely, these same investigators found that use of safety glasses with either a surgical mask or N95 FFR resulted in some eye contamination.The study found a 30% incidence of contamination when using a combination surgical mask with integral eye shield (visor) and 3% for disposable plastic glasses. Utilizing an aerosolized dye (mean particle size 4.8 μm) emitted at a distance of 6 in (15 cm) from subjects wearing two models of face shields, the study noted that the face shields were inferior to two models of surgical face masks tested similarly for particle penetration and that the combination of one of the face-masks with a face shield improved results only marginally. These face shield results were attributable to the lack of a peripheral fit. In a human study using sprayed water during simulated surgery, study observed a 40.5% incidence of contamination of the inner surface of a combination surgical mask with integral visor and 6.5% contamination of the wearers’ face. Study demonstrated that use of a face shield by dental personnel during simulated dental procedures on a mannequin head did not prevent aerosol contamination of a concurrently worn, cup-shaped surgical face mask. Monkey-related Cercopithecine herpesvirus 1 (B virus) infection has been reported in an animal handler and SARS in a nurse, both of whom were wearing a combination surgical mask with integral visor. An epidemiological study reported that the nonuse of face shields by nurses, during high-risk aerosolizing procedures on patients with respiratory infections, resulted in a greater than three-fold increased risk of infection. Use of face shields alone for three months, compared with the use of face masks alone for an equal period, during thoracic and general surgeries resulted in no difference in infection rates of patients. Clearly, there is a need for further research into the protection from infectious airborne pathogens afforded by face shields either worn alone or in conjunction with other PPE worn simultaneously. This should include well-designed aerosol transmission studies,as well as possibly pursuing innovative approaches to design and function (incorporating miniature fans to purge air from the face shield deadspace, application of biostatic films for decontamination purposes, etc.).
Regulatory standards
There is currently no universal standard for face/eye protection from biological hazards. Therefore, the recommendations for the proper use of face shields vary widely, indicating the need for a consensus on the use of certain face/eye protection for specific medical procedures. OSHA’s Bloodborne Pathogens standard (1910:1030 subpart (d)(3)(i)) states: “Masks in combination with eye protection devices, such as goggles or glasses with solid side shields, or chin-length face shields, shall be worn whenever splashes, spray, spatter, or droplets of blood or other potentially infectious materials may be generated and eye, nose, or mouth contamination can be reasonably anticipated.” Face shield product performance specifications are found in the recently-revised voluntary ANSI/ISEA Z87.1–2015 American National Standard for Occupational and Educational Eye and Face Protection that identifies face shields, from an industrial standpoint, as being designed to protect from impact, optical radiation, droplet, and splash (e.g., chemical), dust and fine dust particles, but does not cover bloodborne pathogens, X-rays, high-energy particulate radiation, microwaves, radio-frequency radiation, lasers, masers, and sports and recreation. Face shields do not protect fully from impact hazards, so that OSHA requires their use in conjunction with additional eye protection (goggles, prescription spectacles with side shields, etc.). From the infection control standpoint, no standards currently exist regarding performance standards, but the ISEA Eye and Face Protection Group has initiated development of a voluntary standard that sets forth criteria related to the general performance requirements, test methods, and permanent markings of protectors to minimize or prevent eye and face exposure of the wearer to sprays, splashes, or droplets of blood, body fluids, excretions, secretions, and other potentially infectious materials in occupational and educational environments where biological hazards are expected and routine. Face shields are considered Class I medical devices that are exempt from Food and Drug Administration (FDA) Pre-Market Notification (Form 510[K]), but are subject to the FDA’s Quality System Regulation (21 Code of Federal Regulations 820) that includes periodic inspection and enforcement actions (warning letters, injunctions, seizure, civil monetary penalties). Face masks (surgical, medical, etc.) that incorporate a face shield or visor into their design are considered Class II medical devices and required to submit an FDA Form 510(K).
Guidelines
There is great variance in official (governmental) and professional society (medical, dental, etc.) guidelines for the appropriate use of face shields in the context of protection from biological hazards. Healthcare Infection Control Practices Advisory Committee/CDC Standard Precautions guidelines for prevention of transmission of infectious agents in healthcare venues includes the use of face shields (with a medical/surgical face mask) when sprays, splashes, or splatter are anticipated. The World Health Organization’s health care facility recommendations for standard precautions include a face shield as an alternative to the use of a medical/surgical or procedural mask with eye protection (eye visor or goggles). Similarly, the Ohio State Dental Board guidelines for infection control states that dental healthcare workers need not wear medical/surgical masks when wearing an appropriate face shield that provides protection at both the top and the sides. The Organization for Safety, Asepsis, and Protection, an advocacy group for dental practitioners, advises that use of a face shield alone for protection from contamination by body fluids is likely insufficient, and it is prudent to assume that in those situations where a face shield is used to protect against splash or splatter, a medical/surgical mask would also be indicated. New York State mandatory infection control training offered by approved providers to healthcare workers and medical and physician assistant students states: “When skin protection, in addition to mouth, nose and eye protection, is needed or desired, for example, when irrigating a wound or suctioning copious secretions, a face shield can be used as a substitute to wearing a mask or goggles. The face shield should cover the forehead, extend below the chin, and wrap around the side of the face.” The use of a minimum of an N95 FFR, concurrent with a face shield, has been advocated for protection from serious airborne respiratory infectious diseases (e.g., novel influenza A viruses, SARS) and during procedures on infected persons that result in aerosolization of body fluids (airway suctioning, intubation, etc.). The CDC and U.S. Air Force Dental Evaluation and Consultation Service both promote the use of a medical/surgical mask with a face shield during dental procedures, whereas an Association of Surgical Technologists’ standard on eye protection during surgery mandates the wearing of goggles with face shields during invasive surgical procedures. The ISEA Eye and Face Protection Group has initiated development of a voluntary standard that sets forth criteria related to the general performance requirements, test methods, and permanent markings of protectors to minimize or prevent eye and face exposure of the wearer to sprays, splashes, or droplets of blood, body fluids, excretions, secretions, and other potentially infectious materials in occupational and educational environments where biological hazards are expected and routine.
Selection of face shields
Face shields are meant to be used as barrier protection for the facial area and associated mucous membranes from airborne body fluids (blood, saliva, bronchial secretions, vomit, urine, etc.) expelled as a result of various physiological processes (vomiting, coughing, sneezing, etc.) and medical, dental, and veterinary procedures (suctioning the airway, placing nasogastric tubes, obstetrical procedures, surgery, dental procedures, etc.). Inasmuch as there are currently no standards for face/eye protection against biological hazards, and research data is scant, recommendations for the proper selection of face shields for infection control must rely on currently available knowledge, the task to be performed and the anticipated risk associated with the procedure. The selection of the most appropriate face shield model(s) will depend on the circumstances of exposure, other PPE used concurrently, and personal vison needs.Face shields with single Velcro or elastic straps tend to be easiest to don and doff; doffing can be accomplished with a single hand. In order to be efficacious, face shields must fit snugly to afford a good seal to the forehead area and also to prevent slippage of the device. Visors manufactured from acetate, propionate, and polycarbonate offer improved visual clarity and optical quality with the potential for less eye strain. Visors that offer protection from UV light would be an important feature for individuals utilizing UV light sources (e.g., dental personnel). Face shields should be selected that have visors treated for anti-glare, anti-static, and anti-fogging properties. For improved protection from infectious agents, face shields should be, at a minimum, full face length with outer edges of the face shield reaching at least to the point of the ear, include chin and forehead protectors, and cover the forehead. Brow caps or forehead cushions should be of sufficient dimensions to ensure that there is adequate space between the wearer’s face and the inner surface of the visor to allow for the use of ancillary equipment (medical/surgical mask, respirator, eyewear, etc.). Cost-effective considerations include disposable face shields vs. reusable models and those that offer replaceable parts. Although some models of industrial face shields could be used for infection control purposes (e.g., in the event of face shield shortages), they generally tend to be more expensive, heavier and bulkier than face shields used for infection control purposes.
Proper use of face shields
Correct use of a face shield is dependent upon the indications for use. Appropriately fitted, indirectly vented goggles offer the most reliable practical eye protection from splashes, but face shields are considered an alternative to goggles for prevention of eye contamination with infectious agents. Any additional protection afforded the eyes when protective eyewear (e.g., safety glasses or goggles) is combined with a face shield has not been thoroughly investigated, though the combination of a face shield and goggles has been espoused for use during invasive surgical procedures. The combined use of some forms of protective eyewear with a face shield may impact visual clarity and limit peripheral vison to some extent and these effects must be taken into consideration before use. Goggles have also been reported to fog more than face shields. Also, if a respirator is required in conjunction with face shield use, goggles may not fit properly over the respirator. Use of a face shield alone for eye, face, and mucous membrane protection from contamination by body fluids is likely insufficient and it has been recommended that in those situations where a face shield is used to protect against splash or splatter, a medical/surgical mask would also be indicated. Face shields are not meant to function as primary respiratory protection and should not be used alone because aerosols can flow behind the visor, so a protective facemask (medical/surgical mask, N95 FFR, etc.) should be worn concurrently. In those instances where aerosolization of body fluids of infectious individuals is likely to occur (suctioning the airway, intubation, etc.), a respirator (e.g., N95 FFR, at a minimum) should be used in conjunction with the face shield. Medical/surgical masks with integral visors should not be relied upon as optimal protection, as evidenced by facial and ocular contamination in human and nonhuman research studies and human ocular exposure to infectious agents when wearing these combination devices. The recommended PPE donning and doffing sequence for a face shield in healthcare settings should be followed (donning sequence is gown, protective facemask, face shield [or goggles] and gloves; the doffing sequence is the reverse) keeping in mind that it may vary according to the equipment needed for the particular hazard. Although some models of industrial face shields could be used for infection control purposes (e.g., in the event of face shield shortages), they generally tend to be more expensive, heavier and bulkier than face shields used for infection control purposes.
Face shields are PPE that are commonly used as barrier protection for infection control purposes by numerous workers. There currently is no standard regarding face/eye protection from biological hazards and this deficit needs to be remedied as quickly as possible. Due to the lack of a good facial seal peripherally that can allow for aerosol penetration, face shields should not be used as solitary face/eye protection, but rather as adjunctive to other PPE (protective facemasks, goggles, etc.). Given the dearth of available data regarding the appropriate use of face shields for infection control, scientifically sound research needs to be conducted on the use of this form of PPE
