- IFO380 & IFO180 are Max 3.5% Sulfur Bunkers (RME, RMF, RMG, RMH, RMK)
- LS380 & LS180 are Max 1.0% Sulfur Bunkers
- ULSFO is Max 0.10% Sulfur Fuel Oil for Compliance with 2015 ECA Regulations
- MGO is, unless otherwise specified, a Max 1.50% Sulfur “Clear and Bright” Distillate (DMA, DMZ)
- LSMGO is Max 0.10% Sulfur Distillate (DMA, DMZ) for Compliance with 2015 ECA Regulations
- MDO is Max 1.50% Sulfur Distillate (DMB)
Fuel oil (also known as heavy oil, marine fuel or furnace oil) is a fraction obtained from petroleum distillation, either as a distillate or a residue. In general terms, fuel oil is any liquid fuel that is burned in a furnace or boiler for the generation of heat or used in an engine for the generation of power, except oils having a flash point of approximately 42 °C (108 °F) and oils burned in cotton or wool-wick burners. Fuel oil is made of long hydrocarbon chains, particularly alkanes, cycloalkanes and aromatics. The term fuel oil is also used in a stricter sense to refer only to the heaviest commercial fuel that can be obtained from crude oil, i.e., heavier than gasoline and naphtha.
Small molecules like those in propane, naphtha, gasoline for cars, and jet fuel have relatively low boiling points, and they are removed at the start of the fractional distillation process. Heavier petroleum products like Diesel and lubricating oil are much less volatile and distill out more slowly, while bunker oil is literally the bottom of the barrel; in oil distilling, the only things denser than bunker fuel are carbon black feedstock and bituminous residue (asphalt), which is used for paving roads and sealing roofs.
In Europe, the use of diesel is generally restricted to cars (about 40%), SUVs (about 90%), and trucks and buses (over 99%). The market for home heating using fuel oil, called heating oil, has decreased due to the widespread penetration of natural gas as well as heat pumps. However, it is very common in some areas, such as the Northeastern United States.
Residual fuel oil is less useful because it is so viscous that it has to be heated with a special heating system before use and it may contain relatively high amounts of pollutants, particularly sulfur, which forms sulfur dioxide upon combustion. However, its undesirable properties make it very cheap. In fact, it is the cheapest liquid fuel available. Since it requires heating before use, residual fuel oil cannot be used in road vehicles, boats or small ships, as the heating equipment takes up valuable space and makes the vehicle heavier. Heating the oil is also a delicate procedure, which is impractical on small, fast moving vehicles. However, power plants and large ships are able to use residual fuel oil.
Use of residual fuel oil was more common in the past. It powered boilers, railroadsteam locomotives, and steamships. Locomotives, however, have become powered by diesel or electric power; steamships are not as common as they were previously due to their higher operating costs (most LNG carriers use steam plants, as “boil-off” gas emitted from the cargo can be used as a fuel source); and most boilers now use heating oil or natural gas. Some industrial boilers still use it and so do some old buildings, including in New York City. In 2011 The City estimated that the 1% of its buildings that burned fuel oils No. 4 and No. 6 were responsible for 86% of the soot pollution generated by all buildings in the city. New York made the phase out of these fuel grades part of its environmental plan, PlaNYC, because of concerns for the health effects caused by fine particulates, and all buildings using fuel oil No. 6 had been converted to less polluting fuel by the end of 2015.
Residual fuel’s use in electrical generation has also decreased. In 1973, residual fuel oil produced 16.8% of the electricity in the US. By 1983, it had fallen to 6.2%, and as of 2005, electricity production from all forms of petroleum, including diesel and residual fuel, is only 3% of total production. The decline is the result of price competition with natural gas and environmental restrictions on emissions. For power plants, the costs of heating the oil, extra pollution control and additional maintenance required after burning it often outweigh the low cost of the fuel. Burning fuel oil, particularly residual fuel oil, produces uniformly higher carbon dioxide emissions than natural gas.
Heavy fuel oils continue to be used in the boiler “lighting up” facility in many coal-fired power plants. This use is approximately analogous to using kindling to start a fire. Without performing this act it is difficult to begin the large-scale combustion process.
The chief drawback to residual fuel oil is its high initial viscosity, particularly in the case of No. 6 oil, which requires a correctly engineered system for storage, pumping, and burning. Though it is still usually lighter than water (with a specific gravity usually ranging from 0.95 to 1.03) it is much heavier and more viscous than No. 2 oil, kerosene, or gasoline. No. 6 oil must, in fact, be stored at around 38 °C (100 °F) heated to 65–120 °C (149–248 °F) before it can be easily pumped, and in cooler temperatures it can congeal into a tarry semisolid. The flash point of most blends of No. 6 oil is, incidentally, about 65 °C (149 °F). Attempting to pump high-viscosity oil at low temperatures was a frequent cause of damage to fuel lines, furnaces, and related equipment which were often designed for lighter fuels.
For comparison, BS 2869 Class G heavy fuel oil behaves in similar fashion, requiring storage at 40 °C (104 °F), pumping at around 50 °C (122 °F) and finalizing for burning at around 90–120 °C (194–248 °F).
Most of the facilities which historically burned No. 6 or other residual oils were industrial plants and similar facilities constructed in the early or mid 20th century, or which had switched from coal to oil fuel during the same time period. In either case, residual oil was seen as a good prospect because it was cheap and readily available. Most of these facilities have subsequently been closed and demolished, or have replaced their fuel supplies with a simpler one such as gas or No. 2 oil. The high sulfur content of No. 6 oil—up to 3% by weight in some extreme cases—had a corrosive effect on many heating systems (which were usually designed without adequate corrosion protection in mind), shortening their lifespans and increasing the polluting effects. This was particularly the case in furnaces that were regularly shut down and allowed to go cold, since the internal condensation produced sulfuric acid.
Environmental cleanups at such facilities are frequently complicated by the use of asbestos insulation on the fuel feed lines. No. 6 oil is very persistent, and does not degrade rapidly. Its viscosity and stickiness also make remediation of underground contamination very difficult, since these properties reduce the effectiveness of methods such as air stripping.
When released into water, such as a river or ocean, residual oil tends to break up into patches or tarballs—mixtures of oil and particulate matter such as silt and floating organic matter- rather than form a single slick. An average of about 5-10% of the material will evaporate within hours of the release, primarily the lighter hydrocarbon fractions. The remainder will then often sink to the bottom of the water column.
General Classification
United States
Although the following trends generally hold true, different organizations may have different numerical specifications for the six fuel grades. The boiling point and carbon chain length of the fuel increases with fuel oil number. Viscosity also increases with number, and the heaviest oil must be heated for it to flow. Price usually decreases as the fuel number increases.
Number 1 fuel oil is a volatile distillate oil intended for vaporizing pot-type burners. It is the kerosene refinery cut that boils off immediately after the heavy naphtha cut used for gasoline. Former names include: coal oil, stove oil and range oil.
Number 2 fuel oil is a distillate home heating oil. This fuel is sometimes known as Bunker A. Trucks and some cars use similar diesel fuel with a cetane number limit describing the ignition quality of the fuel. Both are typically obtained from the light gas oil cut. Gas oil refers to the original use of this fraction in the late 19th and early 20th centuries – the gas oil cut was used as an enriching agent for carburetted water gas manufacture.
Number 3 fuel oil was a distillate oil for burners requiring low-viscosity fuel. ASTM merged this grade into the number 2 specification, and the term has been rarely used since the mid-20th century.
Number 4 fuel oil is a commercial heating oil for burner installations not equipped with preheaters. It may be obtained from the heavy gas oil cut.
Number 5 fuel oil is a residual-type industrial heating oil requiring preheating to 77–104 °C (171–219 °F) for proper atomization at the burners. This fuel is sometimes known as Bunker B. It may be obtained from the heavy gas oil cut, or it may be a blend of residual oil with enough number 2 oil to adjust viscosity until it can be pumped without preheating.
Number 6 fuel oil is a high-viscosity residual oil requiring preheating to 104–127 °C (219–261 °F). Residual means the material remaining after the more valuable cuts of crude oil have boiled off. The residue may contain various undesirable impurities, including 2% water and 0.5% mineral soil. This fuel may be known as residual fuel oil (RFO), by the Navy specification of Bunker C, or by the Pacific Specification of PS-400.
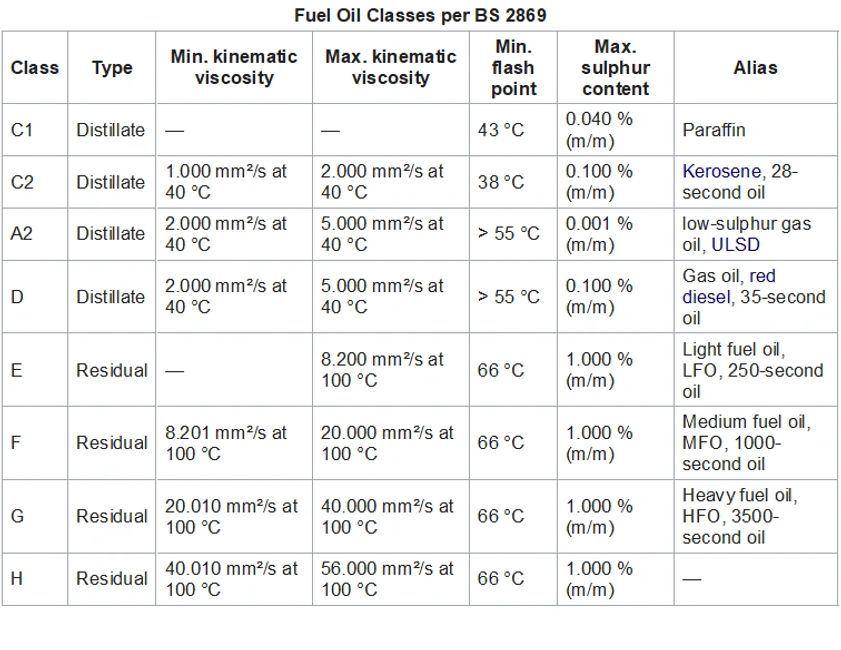
United Kingdom
The British Standard BS 2869, Fuel Oils for Agricultural, Domestic and Industrial Engines, specifies the following fuel oil classes:
Marine Heavy Fuel Oil (HFO) For Ships
The basic requirement for any marine engine is to propel a ship or to generate power onboard by using the energy obtained from burning of fuel oil. HFO or heavy fuel oil is the most widely used type of fuels for commercial vessels.
The fuel oil releases energy to rotate the ship propeller or the alternator by burning fuel inside the combustion chamber of the engine or to generate steam inside the boiler.
The amount of heat energy thus released is the specific energy of a fuel and is measured in MJ/kg.
Under MARPOL Annex 1, the definition of Heavy Grade Oil is given as:
· Crude oils having a density at 15ºC higher than 900 kg/m3;
· Fuel oils having either a density at 15ºC higher than 900 kg/ m3 or a kinematic viscosity at 50oC higher than 180 mm2/s; and
· Bitumen, tar, and their emulsions
History of Marine Heavy Fuel Oil Usage
During the early 19th Century, the cargo ships, which used sails harnessing wind energy, started getting replaced by steamships.
Later on, around the second half of the 20th century, motor ships using IC engines were mainly used as commercial vessels to carry cargo.
The first four-stroke marine engine using heavy fuel was made operational in the 1930s. With time, shipping companies started investing more in R&D and the two-stroke engine became bigger, powerful and famous.
The use of marine heavy fuel oil became more popular in the 1950s because of the introduction of high alkaline cylinder lubrication, which was able to neutralise the acids generated by high sulphur content in the heavy fuel oil.
In the 1960s, ships with marine engine burning heavy fuel oil became more popular and increased in number as compared to the steamships
Eventually, in the 21st century, motor ships replaced almost all steamships and acquired 98% of the world fleet.
What Are The Properties Of Heavy Fuel Oil as per ISO 8217:2010?
Catalytic fines:
Post the refining process; mechanical catalyst particles (aluminium silicate) remain in the oil and are not easy to separate. If exceeded in number, this can damage parts of the fuel system such as an injector, fuel pumps etc. as they have very fine clearance. As per ISO 8217:2010, the maximum limit for Al+Si is 60 mg/kg for RMG and RMK category fuels.
Density:
Every matter, whether solid, liquid or gas has a specific density. The “fuel oil density” is an essential factor that indicates the ignition quality of a fuel and is also used for calculating the amount of fuel oil quantity delivered during the bunkering procedure.
The official and most commonly used unit for density is kg/m3 at 15°C.
Kinematic Viscosity:
Viscosity is the resistance within the fluid which acts against the flow. Kinematic viscosity represents the dynamic viscosity of a fluid per unit density. The viscosity of fuel is a highly significant parameter as it is used to determine the ease of atomization and convenience to pump the fuel within the system.
Calculated Carbon Aromaticity Index (CCAI):
The Calculated Carbon Aromaticity Index (CCAI) is a calculation based on the density and viscosity of a given fuel. As per the formula, the CCAI number is inversely proportional to efficient combustion. This means that higher the CCAI number, the more inferior the ignition quality of the fuel. CCAI helps in getting the ignition delay of the fuel and is used only for the residual fuel such as HFO. The maximum acceptable valve for HFO CCAI is 870.
Flashpoint:
The temperature at which the vapour of the heated fuel ignites is known as the fuel’s flash point. This is done under specified test conditions, using a test flame. As per SOLAS, The flashpoint for all heavy fuel oil to be used onboard vessels is set at Pensky–Martens closed- cup 60°C minimum.
Pour point:
The pour point is the temperature below which the fuel ceases to flow. Once the fuel oil temperature goes below the pour point, it forms wax which can lead to blockage of the filter. The wax formation will also build upon tank bottoms and heating coils, leading to a reduction in heat exchanging capabilities.
Sulphur:
Sulphur in the fuel is one of the main factors for sulphur oxide pollution from ships – a pollutant which is currently under major scrutiny. As per MARPOL, the current sulphur value for HFO are:
· 3.50% m/m on and after 1 January 2012
· 0.50% m/m on and after 1 January 2020
- Water content:
- Water in fuel leads to a decrease in the efficiency of fuel oil and leads to energy loss. Heavy fuel oil mixed with water, if burnt, will lead to corrosion of internal parts.
- Carbon residue:
- A lab test of fuel can determine the carbon residue in the heavy fuel oil. The fuel tends to form carbon deposits on the surface of different parts involved in the combustion chamber under a high-temperature condition. More the amount of hydrocarbons, more difficult to burn the fuel efficiently.
- Ash:
- The amount of inorganic materials present in the fuel which remain as residue once the combustion process is over is called ash deposits. These deposits mainly consist of elements such as vanadium, sulphur, nickel, sodium, silicon, aluminium etc., which are already present in the fuel. The maximum limit of ash content in the fuel is 0.2% m/m.
Problems Of Burning HFO:
1. Water in Fuel: Water in fuel creates issues such as a decrease in heat transfer rate, loss in efficiency and wear of cylinder liner surface etc. Water can mix with fuel oil in different ways such as change of temperature leading to condensation, leaking steam pipe inside the fuel oil tank, improper storage of fuel oil (open sounding pipe) etc.
2. Sludge formation: A ship needs to carry heavy fuel oil in abundance to ensure a continuous supply of fuel to engines and boilers during the long voyage. The heavy fuel oil is stored in the ship’s bunker tanks. Storage of such a large quantity of fuel leads to sludge formation which involves a thick layer on the bottom surface of the tanks. The sludge also sticks on the heat transfer surface of the steam pipes.
3. Pumpability: Many times, if the heating system of the bunker tanks fails or face a problem, it becomes difficult for the ship’s staff to pump the heavy fuel oil from bunker to settling tank due to the high viscosity of the oil. If the heavy fuel oil is of inferior quality, it will choke the filter frequently, increasing the workload of the ship staff onboard ship.
4. Mixing of different grades: Two different grades of heavy oils when mixed together in ship’s storage tanks can lead to stability problems. The number of bunker tanks on ships is limited, and when receiving fuel of different grades, it is a challenge for ship’s officer to store different grades of oils in separate tanks.
5. Combustion: The combustion of heavy fuel oil remains an issue with the ship operator as the oil need to be heated to bring the viscosity below 20cst for achieving proper atomization. If there is an issue in the heating and pumping system, the atomisation will be affected, leading to carbon deposits on the piston and liner surfaces.
6. Abrasion: The heavy fuel oil contains deposits such as vanadium, sulphur, nickel, sodium, silicon etc. which are difficult to remove and have an abrasive effect on the liner and piston surfaces.
7. Corrosion: Elements such as vanadium and sulphur, which are present in the heavy fuel oil, leads to high temperature and low-temperature corrosion respectively.
Vanadium when comes in contact with sodium and sulphur during the combustion, forms a eutectic compound with a low melting point of 530°C.
This molten compound is highly corrosive and attacks the oxide layers on the steel liner and piston (which is used to protect the steel surface), leading to corrosion.
Sulphur is also present in the heavy grade fuel. When sulphur combines with oxygen to form sulphur dioxide or sulphur trioxide, it further reacts with moisture (which can be due to low load operation) to form vapours of sulphuric acid. When the metal temperature is below the dew point of acid, the vapours condense on the surface and cause low-temperature corrosion.
8. Lube oil contamination: During the operation, the heavy fuel oil can always enter the lubrication system and contaminate the lubricating oil. It can be due to leakage through the stuffing box, leaking fuel pumps, or unburned HFO that remains on the cylinder walls and washes down into the sump.
What Are Treatment Methods of Marine Heavy Fuel Oil Used Onboard Ship?
It is impossible to use the heavy fuel oil directly from the bunker tank without treating it. There are different methods used on a ship to treat the fuel before using it for combustion. Some of the most used methods are:
1. Heating & Draining: The fuel delivered to the ship is stored in the bunker tank where it is heated by supplying steam to the coils installed in the bunker tanks. Heating is an essential process, which makes it an integral part of fuel oil treatment. The average temperature maintained for heavy fuel oil bunker tanks is around 40ºC. After transferring it to a settling tank, the fuel is further heated to ensure it is at an appropriate temperature to enter the separators. Once the fuel is transferred to the service tank from the separator, the oil temperature is >80ºC. The main intention is to ensure the smooth pumpability of the fuel oil at different processes and to separate the maximum quantity of water from fuel by draining the settling and service tanks and using purifiers.
2. Purifiers: For removal of water and sludge from the heavy grade oil, fuel oil purifiers are used. Depending upon the owner’s choice, either conventional or modern purifiers (computer driven fuel cleaning systems) can be installed on a ship. The oil flow remains continuous even during the sludge discharge process. Purification of heavy fuel oil is considered to be the most critical treatment process and is carried out on all commercial ships.
3. Filtration: Heating and purification process is used to separate water from the fuel. However, the solid impurities such as fine metal particles which can cause abrasion wear in the fuel system must also be removed. A fine filter is placed in the fuel oil supply line, which blocks the fine metal particles. These are full-flow units and the substance used inside the filters is usually natural, or synthetic fibrous woollen felt material.
4. Chemical treatment: Just like the automotive industry where fuel additives are popular, the maritime industry also uses chemicals in fuels for different jobs; However, this process is not much popular. The main types of residual fuel additives for marine heavy fuel oil are:
• pre-combustion additives such as demulsifiers, dispersants
• combustion improvers
• ash modifiers